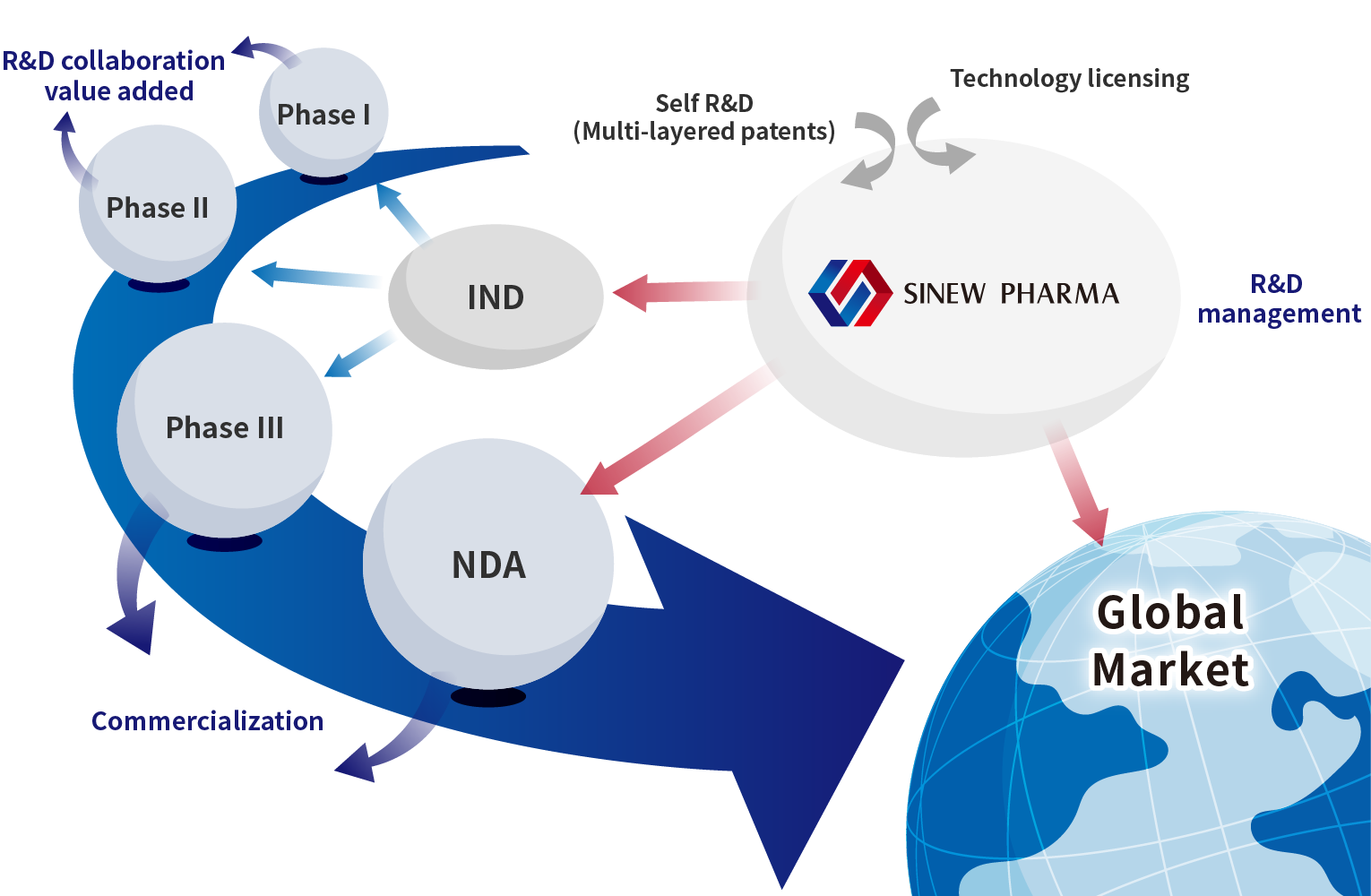
SINEW PHARMA INC. (stock code: 6634.TWO), founded in June 2014, is a publicly-listed biotech company dedicated on the development of therapeutic products for solving urgent and important unmet medical needs.
Leveraging its expertise in three core technologies of "In vitro Co-culture Hepatic Cellular Models," "Hepatic Metabolic Enzyme Activity Regulation Models" and "Pharmacokinetic-driven New Drug Development System”, SINEW is focused on two main disease areas:
- Metabolic dysfunction–associated steatohepatitis (MASH) and its associated conditions, such as liver cirrhosis and liver cancer.
- Acetaminophen-induced hepatotoxicity and severe liver injury.
Driven by the demands in these disease areas, SINEW aspires to become a leading pharmaceutical company by actively engaging in R&D for MASH drug development as well as for the world's first new acetaminophen with high liver safety.
Current pipeline includes six products, two of which are dedicated to MASH treatment, three aim to address liver toxicity issues arising from the widely used painkiller acetaminophen and its combinations, and one serves as an antidote for acetaminophen overdose.